|
12/27/2023 0 Comments Burning candle chemical changeTherefore, burning a candle is a chemical change. The release of heat and light energy is a clear indication of a chemical reaction taking place. Ya burning of candle is chemical change and also physical.when it melts at that time its a physical change and when it threads burn at that time its. The physical changes occur as the wax melts, vaporizes, and is drawn up the wick. 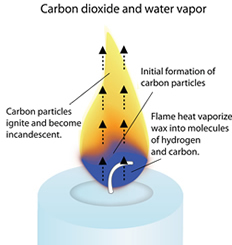
The chemical change occurs as the wax reacts with oxygen, producing carbon dioxide and water vapor. The burning of a candle involves both chemical and physical changes. The gaseous molecules react with oxygen, releasing heat and light energy in the process. The liquid wax vaporizes due to the heat of the flame and turns into a gaseous state. The heat of the flame vaporizes the liquid wax (turns it into a hot gas. 
This liquid wax is then drawn up the wick by capillary action. When you light a candle, the heat of the flame melts the wax near the wick. The solid wax melts and turns into a liquid state, allowing it to be drawn up the wick through capillary action. All waxes are essentially hydrocarbons, which means they are largely composed of hydrogen (H) and carbon (C) atoms. Along with the chemical changes, some physical changes are also observed during the burning of a candle. The carbon atoms in the wax combine with oxygen to produce carbon dioxide (CO2) gas, while the hydrogen atoms combine with oxygen to form water vapor (H2O). These smaller molecules react with oxygen from the air, forming new compounds. In this video I use what you should know about the fire triangle to show you that a candle burning is an example of a chemical change. The heat of the flame then breaks down the hydrocarbon molecules in the vaporized wax into smaller molecules. When the candle is lit, the heat from the flame melts the wax, allowing it to vaporize and rise up the wick. 3.3: Physical and Chemical Changes - Chemistry LibreTexts Combustion & Burning - Energy Foundations. The primary component of a candle is typically a hydrocarbon compound, such as paraffin wax. How burning of candle is physical and chemical change. Because twice as much oxygen is burned than carbon dioxide released, the air volume decreases. For n1 we balance the equation as follows: 2 O 2 + C H 4 C O 2 + 2 H 2 O. The burning produces water H 2 O and carbon dioxide C O 2. This reaction is exothermic, meaning it releases energy. The chemical aspect: oxygen O 2 and paraffin C n H 2n+2 react. Combustion involves a chemical reaction where a substance reacts with oxygen to release energy in the form of heat and light. In the case of a burning candle, the wax undergoes combustion, reacting with oxygen from the air to produce carbon dioxide and water vapor. Chemical changes involve the rearrangement of atoms and the formation of new substances with different properties. And the chemical equation says nothing about the existence of a flame.The burning of a candle is indeed a chemical change. different type waxes have different chemical formulas. When a candle burns, both physical and chemical changes occur: (i) Physical change: melting of wax, vapourisation of melted wax. Give another example of a familiar process in which both the chemical and physical changes take place. Note: Here the chemical formula wax can be different i.e. When a candle burns, both physical and chemical changes take place. Since, it again turns into solid wax on cooling. So, it is a physical change in the burning of candles. So, we can say option D is correct.Īdditional information: On heating, candle wax melts and forms liquid wax. Again the wax combines with the atmosphere oxygen and changes to carbon dioxide, heat and light. Reason: Burning of candle melts the wax and hence physical state of wax has changed from solid to liquid. \įrom the above explanation we can say that formation of new products (carbon dioxide and water) takes place, by the addition of oxygen mass of product will increase and original properties of wax will change. Assertion :The burning of a candle is both a physical and a chemical change. Here, the chemical reaction of burning candle is: Optional: use molecule models to show a simple version of the chemical reaction: CH4 + 2O2 CO2 + 2H2O. Also the properties of the reactants are altered. When a candle is burnt in air, the mass of the product is increased due to atmospheric oxygen which combines with the carbon and hydrogen of the candle wax during burning to produce water vapor and carbon dioxide. 
Burning is a chemical change that involves oxidation thus producing heat and light. The liquid wax evaporates due to the heat produced by the flame. When we light the candle, the wax present near the wick will melt. When we light the candle wax acts as fuel which is a chemical substance called carbon. Hint: Burning of candle is both physical and chemical change.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
